-
Erectile / Penile Health
Erectile Dysfunction Management / Penile Rehabilitation
Erectile Dysfunction Treatment
Rigi10 - Malleable Penile Prosthesis
Infla10 - Inflatable Penile Prosthesis
Restorex for Peyronie's
Next Generation Penile Traction Therapy Device
Restorex for Peyronie's - 3 month data
Restorex for Peyronie's - 6 month data
Restorex - Patient Training - Peyronie's
Penimaster for Peyronies disease
PeniMaster PRO Mode of Operation
Penile Curvature Reduction / Peyronie's Desease Management
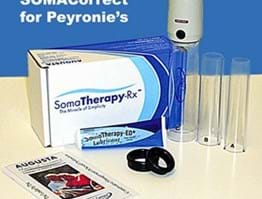
SOMACorrect Vacum Erection Device
PeniMaster PRO Traction Device
Penile Lengthening
PeniMaster PRO - Upgrade Kit II
Premature Ejaculation
Penile Vibrator for Ejaculation (SCI)
Penile Vibrator for Spinal Cord Injured
Viberect® Pro - Penile Vibrator
Intimacy Lubricant
YES Water Based intimacy lubricant
YES Water Based intimacy lubricant - Box of 6 (5ml each)
NOVOGLAN Phimosis Management
Tight Foreskin Phimosis Management Videos
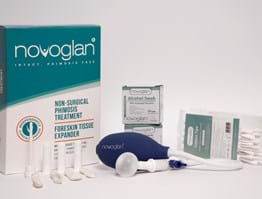
NOVOGLAN Non-Surgical Phimosis Treatment Foreskin Expander Kit
NOVOGLAN Foreskin Conditioning Cream 30ml
NOVOGLAN Foreskin Conditioning Cream 50ml
NOVOGLAN Protective Revitalizing Oil 100ml
NOVOGLAN Refill ( 8 Latex Balloons, 56 Swabs & 56 Buds )
NOVOGLAN Personal Lubricant 100ml
NOVOGLAN Personal Lubricant 50ml
NOVOGLAN Cleansing Liquid Soap 100mL
-
Continence Management / Treatment
Different Types of Incontinence
Maximum Absorbency H-Fly Boxer
Urine Leakage (Moderate)
Maximum Absorbency H-Fly Boxer
Urine Leakage (Severe)
Afex® Male Incontinence System
Urinary incontinence (severe) 24 hours +
Bioderm® Male Continence Device
Cathgrip Catheter and Tube Securement
Cathgrip Single & Double Straps
Urethral Control Devices
Pacey Cuff Powersleeves - pack of three
Pacey Cuff Guard - Reusable Incontinence Pad
Pacey Cuff Comfort Band- pack of three
Urine Leakage Treatment
Incontinence Bedding
-
Incontinence Bedding
-
Diabetic Socks